JOURNEE MONDIALE DE L’AVC : maintenant vous êtes prévenus !
29/10/2015
CANCER : SABLE (E)MOUVANT #PACTONCO
26/11/2015OPEN INNOVATION AT JANSSEN : “Nobody can solve today’s major healthcare problems alone”

OPEN INNOVATION AT JANSSEN : “Nobody can solve today’s major healthcare problems alone”
Located barely an hour’s train ride from Paris, the world’s leading cosmetics and pharmaceutical company Johnson & Johnson has established one of its 14 R&D centres in the town of Val-de-Reuil, Normandy.
As I mentioned in my post, To the pill and beyond… Journey to the heart of Janssen’s innovation, I had the pleasure of visiting the R&D centre which is also a major J&J production site.
My first impression on arrival was the sheer size of the site, which is spread over 27 hectares. This is the second largest J&J site for research and production in Europe.
The Janssen pharmacy R&D centres, along with the Johnson & Johnson and Santé Beauté France cosmetics formulation development centres, are next to the global production facility, surrounded by green grass and fruit trees… It’s a veritable village with over 500 people working in production and 244 across the two research centres !
This site is a centre of excellence within Janssen’s portfolio. So it’s a great opportunity to get a close-up look at innovation in the heart of the company, as a pharmaceutical branch of a global leader in healthcare.
A people-centred culture and Open Innovation: Janssen’s Credo
Around twenty healthcare journalists had come from Spain, the UK, Italy, France, and even Turkey. I was the only blogger in the group, a real privilege!
We were welcomed by Emmanuelle Quilès, President of Janssen France, who shared Janssen’s strategic vision for the years to come.
Firmly anchored in a people-based culture, in particular due to the founding text of the J&J business, the “Credo”, written by the founder in 1943, Janssen is opening up its pharmaceutical laboratory to Open Innovation in all its forms!
Following Emmanuelle Quilès’s talk, we visited the R&D centre with two of the site’s researchers, Jean Michel Argoullon and Jérôme Guillemont.
Jérôme Guillemont received the European Inventor Award 2014 for his discovery in relation to the treatment of multi-resistant tuberculosis.
Jean Michel Argoullon is the operational director of Janssen’s R&D centre.
- Argoullon emphasised that the Val-de-Reuil site is recognised for its expertise in the area of kinases in oncology and in infectious diseases (HIV and multi-resistant tuberculosis). He revealed that each year 5 million euros are invested in external collaborative initiatives with universities, the French National Institute for Agricultural Research (INRA), as well as with biotechs and startups.
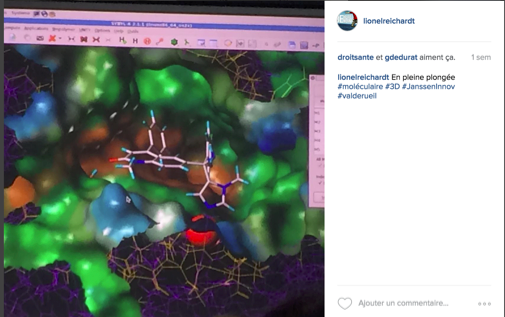
On site, technologies such as 3D printing and digital modelling are used by numerous researchers to understand the mode of action and the interactions between chemical compounds.
We were even given a demonstration. It’s always impressive to travel to the heart of a drug in 3D…
JANSSEN… from essential research to open innovation
This was the title of the round table arranged on site with J&J and Janssen’s managers and partners, all key players in the Open Innovation approach the group has implemented.
This included Janssen Healthcare Innovation (JHI), which launched Care4today in 2012 to improve healthcare results using mobile technology and patient-focused solutions.
“Our team concentrates on areas that can significantly improve the health of customers, such as treatment compliance, cardiac rehabilitation, post-operation recovery and mental health care”, explained Martha Carruthers.
In this way, Janssen Healthcare Innovation works in cross-disciplinary teams to improve the patient experience.
In parallel, Johnson & Johnson founded 4 innovation centres in 2013 in order to strengthen the open innovation strategy, explained Jean Bolger from Johnson & Johnson Innovation.
These centres are situated in 4 strategic locations: London, Boston (on the east coast of the U.S.), California (on the west coast of the U.S.), and Shanghai.
“These innovation platforms are strategically located so that our teams are in the biggest hubs for scientific research in the world.”
Another result of this open innovation approach is the JLABS that J&J has developed to act as ‘no strings attached’ incubators. They provide entrepreneurs with a range of resources: research laboratories fitted with top of the range equipment, shared administrative services, practical working laboratories (known as ‘wet labs’) and offices.
JLABS has 4 sites in the United States (San Diego, South San Francisco, California Institute for Quantitative Biosciences, and Boston and two more will be opened in Houston and Toronto in Canada in 2016).
Ludo Lauwers, Vice Chairman Management Board at Janssen, emphasised that:
“Nobody can solve today’s major healthcare problems alone”.
He even added:
“We have to mix internal research with external research and collaborate with the best centres in the world. We don’t want to just ‘do the basics’ by caring for patients – we want to help them avoid the disease in the first place”.
Ludo Lauwers is also starting work on a new strategy for J&J, which appears to continue this approach: disease interception.
This is a new paradigm, a new approach, in particular in oncology: diagnosing and intercepting cancer in the very first stages, when pre-malignant cells are less complex and less resistant to treatment, patients are in better health and so can get the most from their treatment.
He added that “In the future, healthcare will make use of transformational medicine to prevent and intercept diseases, as well as of the microbiome (the genome of micro-organisms)”.
In February 2015, J&J announced the creation of a new research platform: the Disease Interception Accelerator.
Its mission? To understand how people may be predisposed to a disease by concentrating on its origins, particularly genetic predisposition, environmental exposure and phenotypic changes.
Isn’t the ultimate goal of any scientist to be able to predict the risk of an individual developing a disease before it even manifests itself?
An interesting experience collaborating with EFFIMUNE
Effimune is the first French company to sign an exclusive licence agreement with J&J’s Innovation department, to develop a new medicine for rheumatoid polyarthritis.
Bernard Van Hove, CEO of Effimune, speaks of “a partnership with JANSSEN, where we share the same values”.
Going further, when we met J&J’s consumer products R&D team, we were the first to hear about a project that will demonstrate, in very concrete terms, Janssen’s drive for innovation: In the coming months J&J is going to create a business accelerator and co-working space at the Val-de-Reuil site.
We finished the day with a fascinating visit to the J&J production centre, which is also used for the production of cosmetics and over-the-counter medicines, something that’s quite rare.
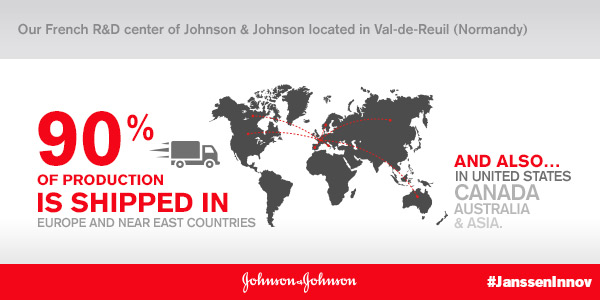
It’s an extremely important site for the group and 90% of its production is exported.
Entirely automated, operating almost ceaselessly round the clock, the site covers 3 floors, and constitutes a veritable hive of activity peppered with vats and stacks of boxes… where everything has been thought of, in the spirit of the excellence and high standards that this type of production demands.
In 2015 the Val-de-Reuil site has also been certified by the FDA, proof of the level of expertise that’s brought to bear on-site.
Your humble servant also paid the price for the high level of excellence and standards required by the production centre as, while Fashion Week was well underway in Paris, he was asked to wear the only outfit permitted for the visit…
Digitally yours,
Lionel REICHARDT – The Pharmageek
Post created in partnership with Janssen France